You searched for:"Sebastião Freitas de Medeiros"
We found (18) results for your search.Summary
Rev Bras Ginecol Obstet. 2004;26(6):463-469
DOI 10.1590/S0100-72032004000600007
OBJECTIVE: to evaluate serum levels of follicle-stimulating hormone (FSH) and luteinizing hormone (LH) in the pill-free interval of a combined oral contraceptive containing 20 mg of ethynylestradiol and 75 mg of gestodene. METHODS: thirty-one women from 17 to 36 years old, mean age of 24.5 years old, 19% adolescents, were included. FSH, LH, prolactin (PRL) and estradiol (E2) levels were measured by immunochemoluminescence. Both FSH and LH levels were measured within the last four days of pill intake and on the 7th day of the pill-free interval between two cycles. Hormonal levels were compared by the Student t-test. Comparisons between hormonal and anthropometric data were made by linear regression; values of p < 0.05 were taken as significant. RESULTS: seventy-one percent of women were using the pill for the first time. FSH levels increased from 1.3 to 5.7 mIU/ml between the end of the blister pack and the 7th day of the pill-free interval. LH increased from 0.8 to 4.3 mIU/ml. E2 levels changed from 20.2 to 28.0 pg/ml. The levels of PRL decreased from 12.4 to 10.2 ng/ml. There was no correlation between the changes in gonadotrophin levels and most of the anthropometric parameters in these women, with body mass index < 25 kg/m². CONCLUSION: the gonadotrophin levels detected on the last four days of pill intake were greatly suppressed, recovery of three to four times in amount occurring on the 7th day of the pill-free interval.
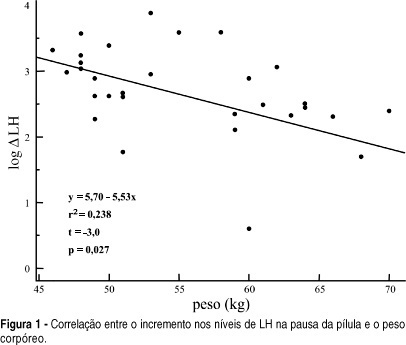
Summary
Rev Bras Ginecol Obstet. 2007;29(1):48-55
DOI 10.1590/S0100-72032007000100008
Changes in the levels of gonadotropins throughout the reproductive life depend on a fine tuned functional development of neural pathways and GnRH-neurones, pituitary gonadotrophs and granulosa-theca cells of the follicular wall. Both, LH and FSH levels change according to the day-time, menstrual cycle phase, and gynecological age. Initiating the puberty, changes in LH pulses are remarkable, showing higher frequency and amplitude at night. Later in puberty, the pulses of LH are also maintained during the day, remaining its levels with very little variation within the 24 hours period. During the menstrual cycle, the FSH levels increase at the end of the luteal phase, decrease during the medium and late follicular phase, increase rapidly in the ovulatory phase and remain at low basal levels until the late luteal phase. The levels of LH remain unaltered during the whole follicular phase, increase in the ovulatory surge, and decrease to the basal levels in the luteal phase. At the forth decade of life, the GnRH secretion changes, with hypothalamic loss of sensitivy to the estradiol positive feedback and decrease in frequency and prolongation of the GnRH pulses. The pituitary response is atenuated due to decrease in the density of GnRH receptors on gonadotroph cells, loss of gonadotroph sensitivity, secretion of more basic FSH and LH molecules, decrease in frequency and increase in amplitude of LH and FSH pulses. These modifications result in monotropic increase of the FSH secretion. Current studies show that the selective increase in the FSH levels in the early follicular phase is gradual, beginning as early as the third decade of life. These alterations in FSH are associated with an accelerated follicular depletion in women after 37-38 years old. On the other side, the LH levels remain almost constant up to the end of reproductive life. The different levels of FSH and LH seen throughout the reproductive years may be due to yet unknown regulatory mechanisms in the hypothalamic-pituitary-ovarian axis.
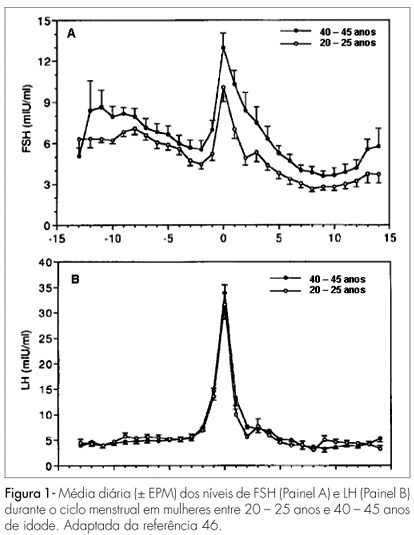
Summary
Rev Bras Ginecol Obstet. 2021;43(6):480-486
The process of ovulation involves multiple and iterrelated genetic, biochemical, and morphological events: cessation of the proliferation of granulosa cells, resumption of oocyte meiosis, expansion of cumulus cell-oocyte complexes, digestion of the follicle wall, and extrusion of the metaphase-II oocyte. The present narrative review examines these interrelated steps in detail. The combined or isolated roles of the folliclestimulating hormone (FSH) and luteinizing hormone (LH) are highlighted. Genes indiced by the FSH genes are relevant in the cumulus expansion, and LH-induced genes are critical for the resumption ofmeiosis and digestion of the follicle wall. A nonhuman model for follicle-wall digestion and oocyte release was provided.
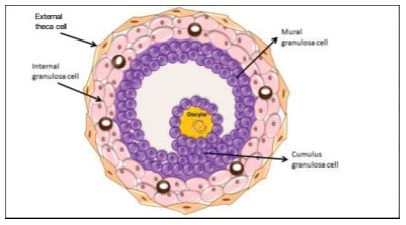
Summary
Rev Bras Ginecol Obstet. 2005;27(9):509-514
DOI 10.1590/S0100-72032005000900002
PURPOSE: to evaluate the luteal function in adolescents with regular menstrual cycles. METHODS: this prospective cohort study included 55 adolescents, aged 14-19 years, with menarche at 12.2 years. Ovulation was identified by ultrasound, starting on the second or fifth day of the cycle. The corpus luteum vascularization and the resistence index of the ovarian vessels were measured by Doppler on the tenth postovulatory day. Progesterone was measured by chemoluminescence on days 6, 9 and 12 of the luteal phase. The endometrial biopsy was performed 8 to 10 days after ovulation. The results were analyzed using the SPSS software and were considered significant when p<0.05. RESULTS: on average ovulation was on day 17. Progesterone levels were 11.4, 10.9 and 3.9 ng/mL on days 6, 9, and 12 after ovulation, respectively; the progesterone mean during the whole luteal phase was 10.3 ng/ml. Luteal vascularization was scarce in 34.6%, mild in 23.6% and exuberant in 41.8%. The resistance index was 0.441. On the tenth day post-ovulation the endometrium was normal in 85.5% and out-of-phase in 14.5%. There was no correlation between the ovulation day and endometrial dating (p=0.294), levels of progesterone and endometrial dating (p=0.454), progesterone and corpus luteum vascularization (p=0.994), or resistance index (p=0.237). There also was no association between endometrium development and degree of vascularization (p=0.611). CONCLUSION: abnormal luteal function in adolescents with regular menstrual cycles was found in 14.5%. Degree of vascularization, resistance index, and serum progesterone were not related to endometrium development.
Summary
Rev Bras Ginecol Obstet. 2010;32(11):541-548
DOI 10.1590/S0100-72032010001100005
PURPOSE: to reassess the adrenal function of patients with PCOS after the introduction of the Rotterdam's criteria. METHODS: descriptive and cross-sectional study including 53 patients 26±5.1 years old. Glucose, glycosylated hemoglobin, lipids, estradiol, progesterone, 17-OHP4, DHEAS, FSH, LH, TSH, PRL, androstenedione, free thyroxine, insulin, total testosterone, SHBG, and free androgen index were measured. Insulin resistance was considered to be present with a homeostatic model assessment index >2.8. The adrenal response to cortrosyn was assessed by the hormonal rise observed at 60 minutes, and by the area under the response curve. RESULTS: biochemical hyperandrogenism was found in 43 of 53 eligible patients (81.1%). Thirty-three women had adrenal hyperandrogenism (62.2%). The weight of these 33 women, aging 25.1±5.0 years, was 74.9±14.9 kg, BMI was 28.8±6.0 and the waist/hip ratio was 0.8±0.1. DHEAS was >6.7 nmol/L in 13 (39.4%) and androstenendione was >8.7 nmol/L in 31 (93.9%). The increments in 17-OHP4, cortisol, A, and progesterone were 163%, 153%, 32%, and 79%, respectively. The homeostatic insulin resistance model was >2.8 in 14 (42.4%). Insulin and estradiol were not correlated with cortisol or androgens. CONCLUSIONS: the use of multiple endocrine parameters showed a high prevalence of biochemical hyperandrogenism in patients with PCOS. Two thirds of the patients had adrenal hyperandrogenism, and estradiol and insulin did not influence adrenal secretion.
Summary
Rev Bras Ginecol Obstet. 2021;43(7):578-578
Summary
Rev Bras Ginecol Obstet. 2008;30(12):591-593
Summary
Rev Bras Ginecol Obstet. 2007;29(11):593-601
DOI 10.1590/S0100-72032007001100008
There is evidence that estrogen, progesterone and testosterone have modulatory effects over both cellular and humoral immune responses. These effects occur via immune-neuroendocrine interactions, involving the pituitary, gonadal steroids, thymic hormones, and the presence of specific receptors and messengers. These immune responses may be altered during pregnancy, gonadectomy, menopause and hormone therapy. Estrogen depresses the cellular immunity, suppresses the natural killer cell activity and increases the production of antibodies. Progesterone/progestogen suppresses the cellular immune system. Androgens, after metabolization in estrogens, might stimulate the humoral immune response. Hormone therapy is still broadly used in post-menopause women with the purpose of decreasing climacteric symptoms, as well as preventing genital atrophy and bone loss. Its use to attenuate the risk of cardiovascular and neurodegenerative diseases remains in debate. A few studies have been carried out to examine the effect of post-menopause hormone therapy on the immune system. There is evidence that the hypoestrogenic state, following menopause, could result in less resistance to infections. The present review examines the interaction between sexual steroids and the immune system and, based on epidemiological and clinical studies, evaluates the effects of hormone therapy on the immune responses. It was concluded that the hormone therapy normalizes the cellular immune response in post-menopausal women.