You searched for:"Ana Katherine Gonçalves"
We found (10) results for your search.Summary
Rev Bras Ginecol Obstet. 2023;45(12):808-817
To assess the efficacy, safety, and acceptability of misoprostol in the treatment of incomplete miscarriage.
The PubMed, Scopus, Embase, Web of Science, Cochrane Library, and Clinical Trials databases (clinicaltrials.gov) were searched for the relevant articles, and search strategies were developed using a combination of thematic Medical Subject Headings terms and text words. The last search was conducted on July 4, 2022. No language restrictions were applied.
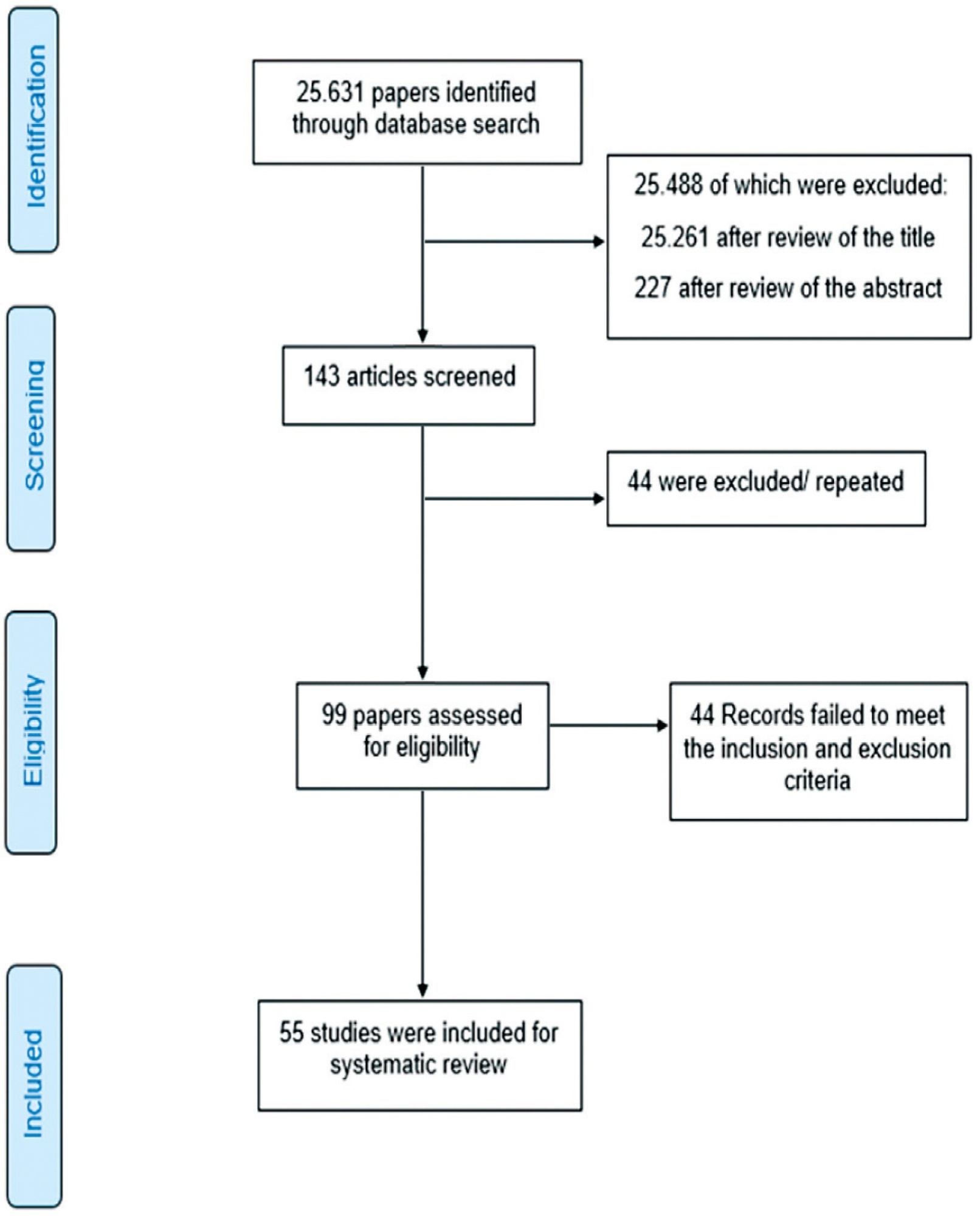
Randomized clinical trials with patients of gestational age up to 6/7 weeks with a diagnosis of incomplete abortion and who were managed with at least 1 of the 3 types of treatment studied were included. A total of 8,087 studies were screened.
Data were synthesized using the statistical package Review Manager V.5.1 (The Cochrane Collaboration, Oxford, United Kingdom). For dichotomous outcomes, the odds ratio (OR) and 95% confidence interval (CI) were derived for each study. Heterogeneity between the trial results was evaluated using the standard test, I2 statistic.
When comparing misoprostol with medical vacuum aspiration (MVA), the rate of complete abortion was higher in the MVA group (OR = 0.16; 95%CI = 0.07–0.36). Hemorrhage or heavy bleeding was more common in the misoprostol group (OR = 3.00; 95%CI = 1.96–4.59), but pain after treatment was more common in patients treated with MVA (OR = 0.65; 95%CI = 0.52–0.80). No statistically significant differences were observed in the general acceptability of the treatments.
Misoprostol has been determined as a safe option with good acceptance by patients.
Summary
Rev Bras Ginecol Obstet. 2022;44(10):986-994
To evaluate the efficacy of the hormonal and nonhormonal approaches to symptoms of sexual dysfunction and vaginal atrophy in postmenopausal women.
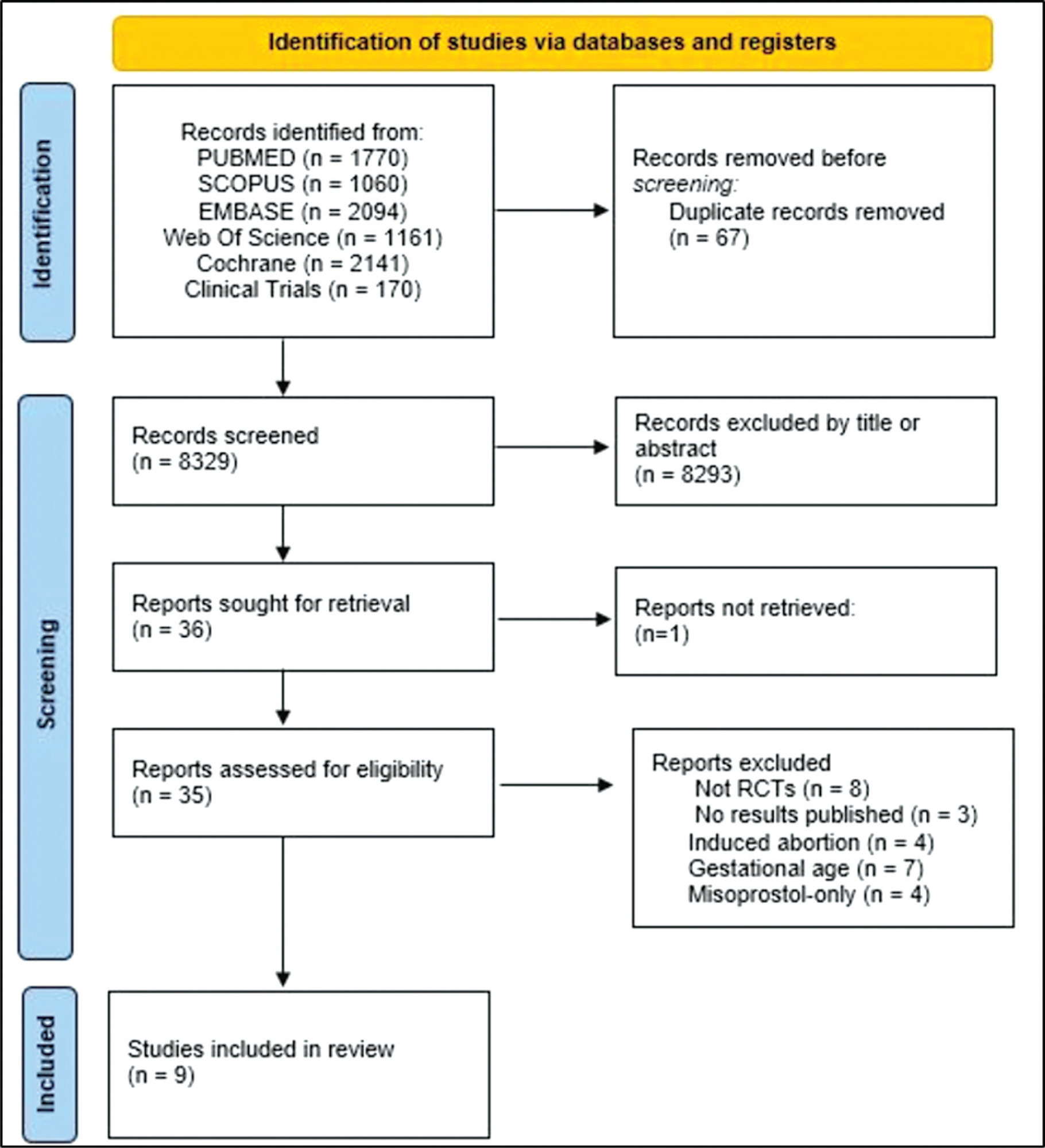
We conducted a search on the PubMed, Embase, Scopus, Web of Science, SciELO, the Cochrane Central Register of Controlled Trials (CENTRAL), and Cumulative Index to Nursing and Allied Health Literature (CINAHL) databases, as well as on clinical trial databases. We analyzed studies published between 1996 and May 30, 2020. No language restrictions were applied.
We selected randomized clinical trials that evaluated the treatment of sexual dysfunction in postmenopausal women.
Three authors (ACAS, APFC, and JL) reviewed each article based on its title and abstract. Relevant data were subsequently taken from the full-text article. Any discrepancies during the review were resolved by consensus between all the listed authors.
A total of 55 studies were included in the systematic review. The approaches tested to treat sexual dysfunction were as follows: lubricants and moisturizers (18 studies); phytoestrogens (14 studies); dehydroepiandrosterone (DHEA; 8 studies); ospemifene (5 studies); vaginal testosterone (4 studies); pelvic floor muscle exercises (2 studies); oxytocin (2 studies); vaginal CO2 laser (2 studies); lidocaine (1 study); and vitamin E vaginal suppository (1 study).
We identified literature that lacks coherence in terms of the proposed treatments and selected outcome measures. Despite the great diversity in treatment modalities and outcome measures, the present systematic review can shed light on potential targets for the treatment, which is deemed necessary for sexual dysfunction, assuming that most randomized trials were evaluated with a low risk of bias according to the Cochrane Collaboration risk of bias tool. The present review is registered with the International Prospective Register of Systematic Reviews (PROSPERO; CRD42018100488).