Summary
Revista Brasileira de Ginecologia e Obstetrícia. 2009;31(5):263-263
Summary
Revista Brasileira de Ginecologia e Obstetrícia. 2001;23(4):263-263
DOI 10.1590/S0100-72032001000400014
Summary
Revista Brasileira de Ginecologia e Obstetrícia. 2016;38(6):263-265
Summary
Revista Brasileira de Ginecologia e Obstetrícia. 2005;27(5):263-267
DOI 10.1590/S0100-72032005000500006
PURPOSE: to evaluate the influence of hyperthyroidism during pregnancy and the necessity of changing antithyroid drug dose in this period and after delivery. METHODS: prospective evaluation of clinical and laboratorial findings of thirteen pregnancies in eleven pregnant women with hyperthyroidism due to Graves' disease. These women were evaluated through TSH and serum free T4 at each trimester or four weeks after setting thionamide dosage. The goal was to maintain free T4 in the superior third of the normal range using the lowest possible thionamide dose. RESULTS: the mean age at the beginning of the pregnancy was 31.1 years (23 to 41). The mean dosage of thionamide was reduced in eight pregnancies (69.5%) and, in two, the drug was discontinued. Before pregnancy, mean propylthiouracil dose was 400 mg/day (200-900) and mean methimazole dose was 45 mg/day (20-60). After delivery, antithyroid drug dose was 200 and 30 mg/day, respectively. One patient presented premature labor (at 36 weeks) and another, a newborn small for gestational age (2.000 g at 38 weeks). There was one stillborn. There were no miscarriages or congenital anomalies. After labor, antithyroid drug dose was increased in seven patients and in the others the dose was maintained. CONCLUSIONS: we suggest close follow-up of pregnant women with hyperthyroidism and progressive reduction of thionamide dose during pregnancy to avoid maternal hypothyroidism and its consequences to fetal development. After labor, these women must be evaluated regarding their thyroid function because hyperthyroidism can worsen. Thionamide use is safe for the patients and their offspring.
Summary
Revista Brasileira de Ginecologia e Obstetrícia. 2001;23(4):264-264
DOI 10.1590/S0100-72032001000400015
Summary
Revista Brasileira de Ginecologia e Obstetrícia. 2022;44(3):264-271
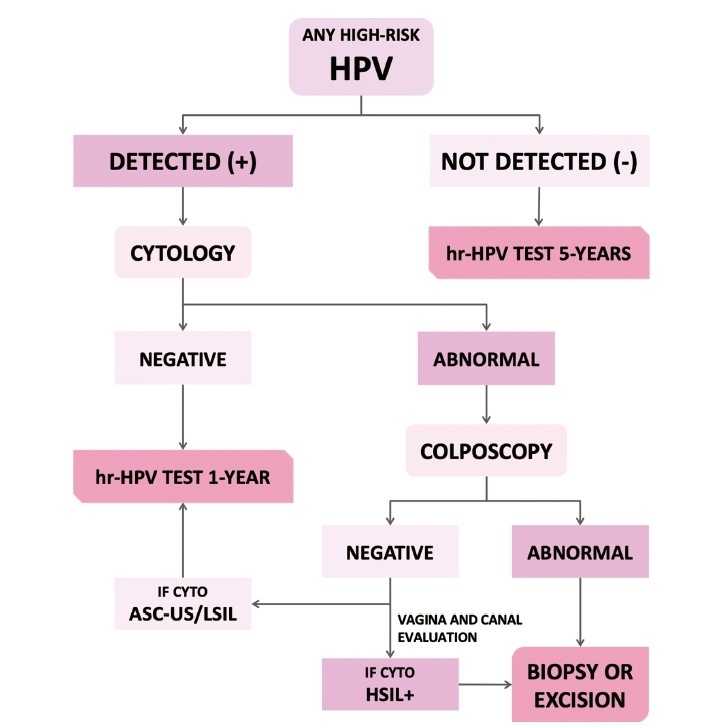
The present update is a reassessment of the 2018 ‘Guidelines for HPV-DNA Testing for Cervical Cancer Screening in Brazil’ (Zeferino et al.)9, according to the changes observed in new international guidelines and knowledge updates. The most relevant and recent guidelines were assessed. Questions regarding the clinical practice were formulated, and the answers considered the perspective of the public and private sectors of the Brazilian health system. The review addressed risk-based strategies regarding age to start and stop screening, the use of cytology and colposcopy to support management decisions, treatment, follow-up strategies, and screening in specific groups, including vaccinated women. The update aims to improve the prevention of cervical cancer and to reduce overtreatment and the misuse of HPV testing.
Summary
Revista Brasileira de Ginecologia e Obstetrícia. 2014;36(6):264-268
DOI 10.1590/S0100-720320140004935
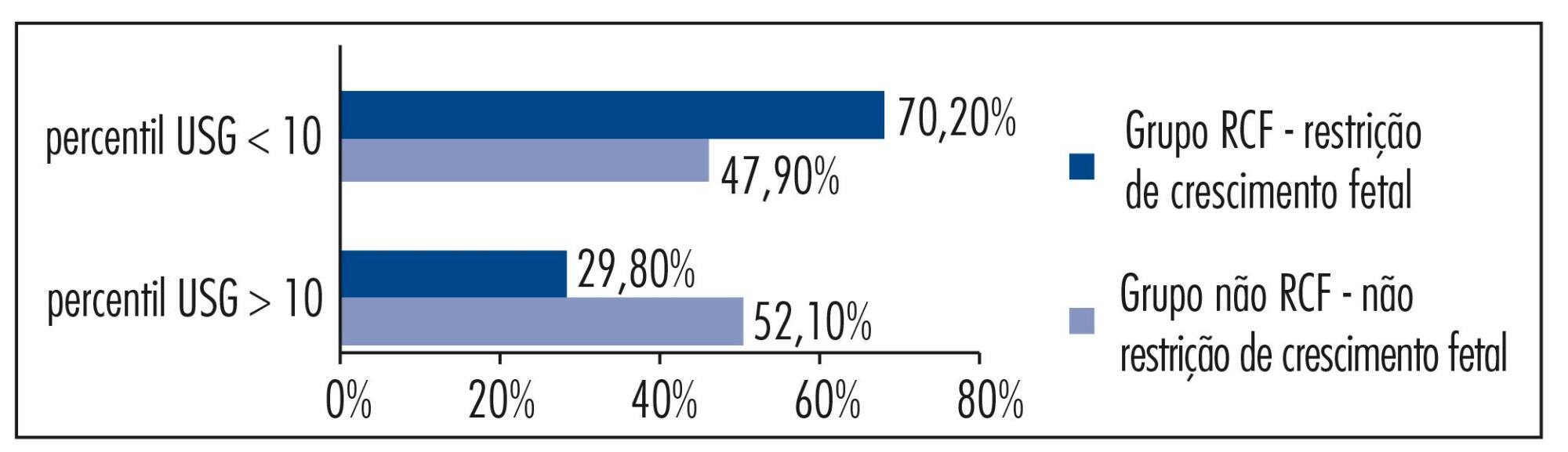
The aim of this study was to analize and describe some characteristics related to a false diagnosis of intrauterine growth restriction (IUGR).
We retrospectively included 48 pregnant women referred to our service with a suspected diagnosis of IUGR that was not confirmed after birth and we compared them to another group with confirmed IUGR. We then analyzed the characteristics of the false-positive results. The results of the study were divided into continuous and categorical variables for analysis. The χ2test or Fisher exact test was applied to compare proportions. The level of significance was set at p<0.05 for all tests.
In our sample, pregnant women with a false diagnosis of IUGR had the following characteristics: they were referred earlier (mean gestational age of 32.8 weeks); were submitted to 2 to 6 ultrasound examinations before been registered in our service; in 25% of cases ultrasound examination was performed before 12 weeks; in 66.7% of cases the symphysis-fundal height measurement was normal; in 52.1% of cases they had at least 1 sonographic exam above the 10th percentile; on average, the last ultrasound examination (performed on average at 36 weeks) was above the 18th percentile; the women were submitted to a mean number of 5 ultrasound examinations and to a mean number of 4.6 vitality exams.
The false diagnosis of IUGR involves high hospital costs and higher demand for specialists. The symphysis-fundal height measurement must be valued, and the diagnosis of IUGR must be confirmed with ultrasonography in the last weeks of pregnancy before any obstetric management is taken.