Revista Brasileira de Ginecologia e Obstetrícia. 2006;28(7):424-430
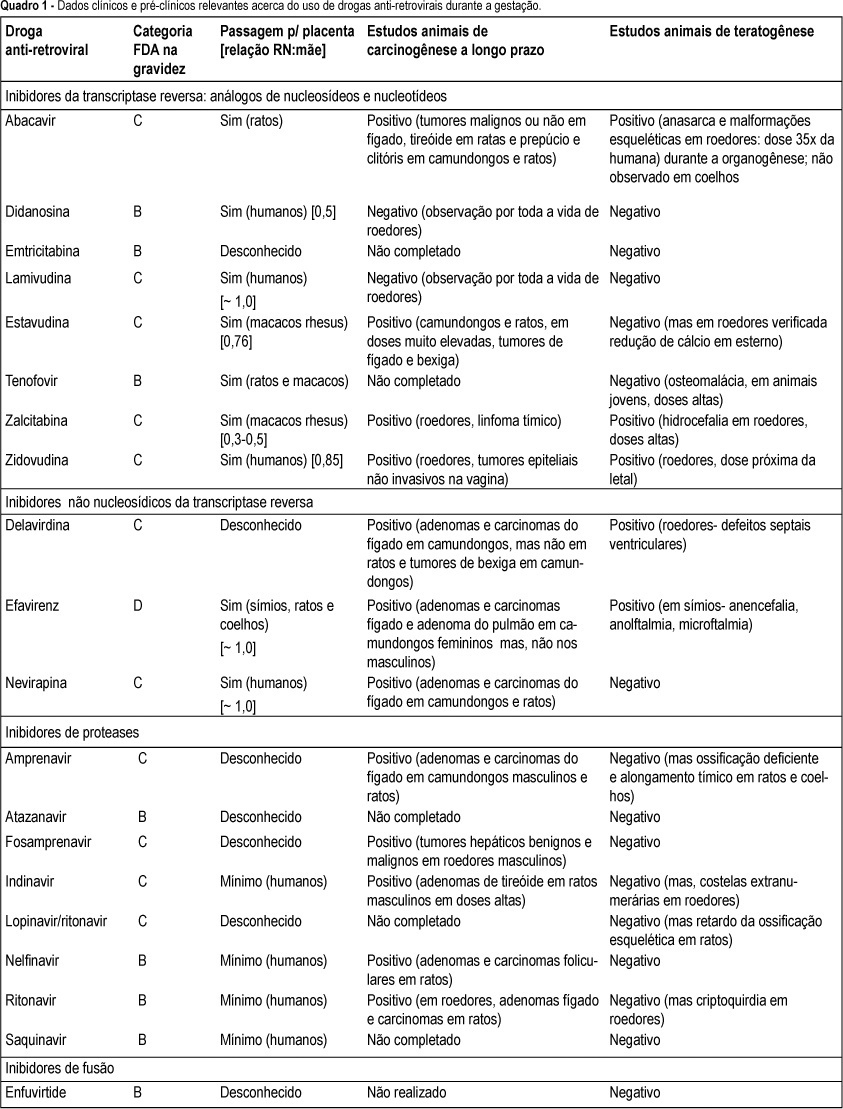
The implementation of prophylactic antiretroviral drugs and others strategies to prevent vertical HIV transmission in Brazil has reduced the number of newly HIV-infected children. Since these drugs are only recently available for treatment, there is no data of conclusive safety during pregnancy and fetus and newborn. This article reviews the possible effects of these drugs in fetus, newborn and childhood, including teratogenesis, carcinogenesis and toxicity. In the literature there are an increasing description of adverse effects such as mithocondrial toxicity, neurological symptoms, blood and liver toxicity in HIV-exposed not infected children. Antiretroviral treatment recommendations are based on principles that known benefit to the pregnat woman should be mantained unless adverse effects outweight the benefit. A long term follow up of HIV- exposed children is proposed to better understanding of these potential toxicity. In this review the author suggests a practical clinical and laboratory routinary evaluation during the first years of life until adolescence for HIV-exposed not infected children.
Search
Search in:


Comments